


Chromium(III) – coordination chemistry
Chromium(III) ion is known to show a very rich coordination chemistry in aqueous solution. In this experiment only very basic chemicals are used and with these chemicals a few not so well-known properties of chromium(III) ion in aqueous solution are demonstrated.

A solution of a simple and easy to obtain chromium(III) salt is prepared and simply heating already gives interesting behavior. When ammonia is added to the game, things become even more interesting.
![]()
![]() Required
chemicals:
Required
chemicals:
-
chrome alum (chromium(III)sulfate also can be used)
-
ammonia (5% and 12%)
-
optional: hydrochloric acid (10%)
-
optional: sodium hydroxide
![]() Required
equipment:
Required
equipment:
-
heat resistant test tube
- bunsen burner or blow torch
![]() Safety:
Safety:
- Ammonia is fairly corrosive and dangerous for the eyes. At the used concentrations some irritating pungent smell may be produced. Do the experiment in a well-ventilated room.
![]() Disposal:
Disposal:
- Only small amounts of trivalent chromium are used in this experiment and this waste may be flushed down the drain with a large amount of water.
![]()
Formation of sulfato-complex by simply heating
![]() Dissolve
a small amount of chrome alum or chromium(III)sulfate in distilled water, such
that a solution is obtained which is not too dark. If the solution is too
concentrated, then the visual effects will not nearly be as nice as described in
this page. Divide the solution over two test tubes. One should start with two
tubes with solution like the following:
Dissolve
a small amount of chrome alum or chromium(III)sulfate in distilled water, such
that a solution is obtained which is not too dark. If the solution is too
concentrated, then the visual effects will not nearly be as nice as described in
this page. Divide the solution over two test tubes. One should start with two
tubes with solution like the following:


One picture is made under TL-light, the other is made under a gray winter sky. The color of the solution is hard to tell precisely, it depends on the light source, but it should be somewhere in the range blue/violet/purple.
![]() Heat the
solution in one of the two test tubes, set the other test tube aside. The
heating must be done slowly and the liquid must be boiled gently for
a minute or so. After the heating, the liquid in the test
tube will have a nice green color. On cooling down, the liquid remains green.
Heat the
solution in one of the two test tubes, set the other test tube aside. The
heating must be done slowly and the liquid must be boiled gently for
a minute or so. After the heating, the liquid in the test
tube will have a nice green color. On cooling down, the liquid remains green.


The green color persists for a very long time. It takes weeks before the color of the liquid reverts to the original blue/violet/purple.
![]()
Formation of ammine-complex
![]() To the
two test tubes, shown above, add equal volumes of 5% ammonia, which is in large
excess, compared to the amount of chromium. After addition of the ammonia, the
test tubes should be shaken. This results in formation of a grayish
precipitates, which only settle very slowly. The precipitates are slimy and
voluminous. The color of the precipitates differs for both test tubes. The green
liquid gives a more bluish precipitate, the blue/violet/purple solution gives a
greyish/green precipitate.
To the
two test tubes, shown above, add equal volumes of 5% ammonia, which is in large
excess, compared to the amount of chromium. After addition of the ammonia, the
test tubes should be shaken. This results in formation of a grayish
precipitates, which only settle very slowly. The precipitates are slimy and
voluminous. The color of the precipitates differs for both test tubes. The green
liquid gives a more bluish precipitate, the blue/violet/purple solution gives a
greyish/green precipitate.

![]() Let the
test tubes stand on a quiet place, such that the precipitates can settle. After
a few hours, the test tubes look as follows:
Let the
test tubes stand on a quiet place, such that the precipitates can settle. After
a few hours, the test tubes look as follows:
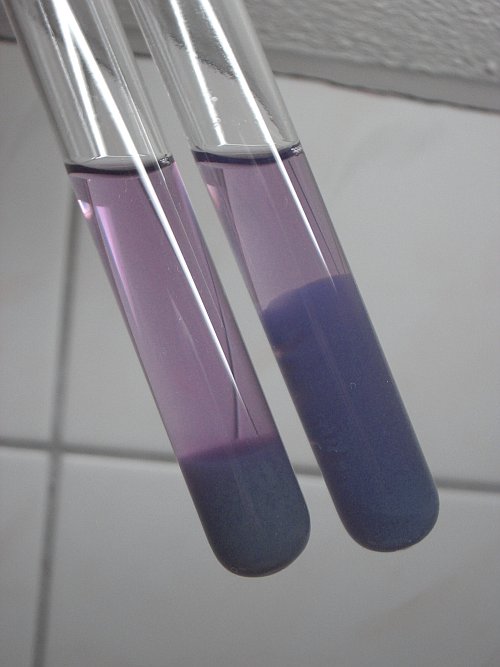
Both test tubes have a pink/purple clear liquid above a slimy precipitate. Still, both precipitates have different colors and one of the precipitates settled more to the bottom than the other.
![]()
Formation of ammine-complex by boiling
In a final experiment, it is shown how a beautiful purple solution can be made from a chrome alum or chromium(III)sulfate. This can be done by making a precipitate of chromium(III) in fairly concentrated ammonia and then boiling.
![]() First, prepare a solution of chrome alum or chromium(III)sulfate, which may look
rather dark. To this solution add a large excess of 12% ammonia. This results in
a grayish precipitate. The precipitate does not redissolve in excess ammonia.
First, prepare a solution of chrome alum or chromium(III)sulfate, which may look
rather dark. To this solution add a large excess of 12% ammonia. This results in
a grayish precipitate. The precipitate does not redissolve in excess ammonia.


![]() Carefully and gently boil the solution for several minutes. When this is done,
then the color of the liquid slowly changes from grayish to nice purple while
still being turbid. The precipitate does not seem to dissolve, but the change of
color of the liquid clearly shows that some of the precipitate dissolves and a
complex is formed with the ammonia in the solution. Formation of the complex
with ammonia is not fast and complete.
Carefully and gently boil the solution for several minutes. When this is done,
then the color of the liquid slowly changes from grayish to nice purple while
still being turbid. The precipitate does not seem to dissolve, but the change of
color of the liquid clearly shows that some of the precipitate dissolves and a
complex is formed with the ammonia in the solution. Formation of the complex
with ammonia is not fast and complete.
After several hours, the precipitate has settled at the bottom. This only settles very slowly. A bright purple liquid remains.

Optional: showing tight binding of ammonia ligands
When the purple solution is added to an excess amount of hydrochloric acid, then the color of the solution does not change. Even in the presence of the strong acid HCl, the ammonia molecules, which are connected to the chromium atoms, are not converted to ammonium ions. This is in strong contrast with the royal blue copper-ammine complex, which immediately is destroyed on addition of acid.
Similarly, when the purple solution is added to excess NaOH, then the solution also remains purple. The ammonia molecules are not stripped from the chromium, such that it forms green chromite-ion.
Both of these final experiments show that the ammonium molecules are bound to the chromium atom very tightly.
![]()
Discussion of results
Chromium(III) is known to form a plethora of complexes in aqueous solution. Virtually every ion or molecule that is capable of forming complexes with transition metal ions will do so with chromium(III). The formation of the complexes, however, is not fast. Ligands, once attached to chromium ions, are coordinated to the metal ion rather tightly and it takes quite some effort to disconnect them from the metal ion and to replace them by something else. This is in strong contrast with e.g. copper(II) ions, which also forms many complexes, but those complexes are labile and ligands easily are ripped from the central metal ion.
![]() In chrome alum or hydrated chromium(III)sulfate, the
chromium ion is present as the hexaaqua ion: Cr(H2O)63+.
When these solids dissolve, then the ion remains present as the hexaaqua ion.
This ion has a somewhat hard to describe color, somewhere in the range
blue/violet/purple with a grayish hue, depending on the type of light, falling
on the solution. Solid salts, containing hexaaqua chromium(III) ions are dark
purple/violet or gray/violet when crushed to a fine powder.
In chrome alum or hydrated chromium(III)sulfate, the
chromium ion is present as the hexaaqua ion: Cr(H2O)63+.
When these solids dissolve, then the ion remains present as the hexaaqua ion.
This ion has a somewhat hard to describe color, somewhere in the range
blue/violet/purple with a grayish hue, depending on the type of light, falling
on the solution. Solid salts, containing hexaaqua chromium(III) ions are dark
purple/violet or gray/violet when crushed to a fine powder.
When such a solution, containing Cr(H2O)63+ and SO42- is heated, then there is slow ligand exchange and molecules of water are replaced by sulfate ions, which mostly act as monodentate ligands in this situation:
Cr(H2O)63+ + SO42- → [Cr(H2O)5SO4]+ + H2O
Exchange can go further and more water molecules can be replaced. It might also be that one sulfate ion acts as a bidentate ligand and then it replaces two water molecules. The precise nature of the sulfate-complexes in solution cannot be described, only a general outline, like the one given here, is possible. These sulfato-complexes have a green color.
When a solution of chromium(III)sulfate or chrome alum is boiled down, then finally one ends up with a green syrup. When this is allowed to cool down, then a glassy green substance is obtained, which is very hard to crystallize. The reason for this is that the substance is not a well-defined entity with a clear structure, but it is a complicated mix of different sulfato-complexes with varying amount of water, depending on how much it was evaporated.
![]() When excess ammonia is added to a solution, containing chromium(III) salts, then
extensive hydrolysis occurs, and a hydrous precipitate of chromium(III)
hydroxide is formed:
When excess ammonia is added to a solution, containing chromium(III) salts, then
extensive hydrolysis occurs, and a hydrous precipitate of chromium(III)
hydroxide is formed:
NH3 + H2O ↔ NH4+ + OH–
Cr(HOH)63+ + 3OH– → Cr(HOH)3(OH)3 + 3HOH
The hexaaqua chromium(III) ion is fairly acidic and it easily donate a few protons. When ammonia is added, then so much hydroxide is formed, that a neutral complex is formed with three hydroxide ligands, and this precipitates as slimy and voluminous hydrous chromium(III)hydroxide.
When there is excess ammonia, then there is not sufficient hydroxide in solution to allow more than three protons to be donated, such that an anionic species is formed from the chromium(III) complex. This does happen in solutions of sodium hydroxide, but ammonia is not sufficiently basic to redissolve the precipitate. With ammonia, however, there is slow ligand exchange with water and with hydroxide, resulting in ammine-complexes of chromium. This can go up all the way to Cr(NH3)63+ and if this occurs, then the chromium goes into solution again. The color of this complex ion is bright purple.
Some background info about this subject can be found here:
- http://www.chemguide.co.uk/inorganic/transition/chromium.html
- http://www.chemguide.co.uk/inorganic/complexions/acidity.html