


Copper acetate & Co.
Copper acetate is not a simple salt, consisting of copper(II) ions and acetate ions, but it is a complex. Usually, (CH3COO)2Cu·H2O is given as the net formula. However, the true formula of copper(II) acetate is (CH3COO)4Cu2(H2O)2. The structure of this compound is shown below.
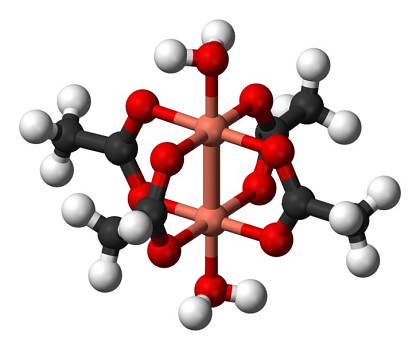
In this structure, all bonds between oxygen atoms and the two central copper atoms are coordinate bonds, where a lone pair of electrons on the oxygen atoms are shared with the copper atoms. The acetate ions are attached to both copper atoms, one oxygen is connected to one copper atom and the other oxygen is connected to the other copper atom.
Macroscopically, one also can clearly see that copper(II) acetate is not a plain copper(II) salt with simple hydrated copper ions. It has a deep green/blue color instead of the well-known sky-blue color of ordinary hydrated copper(II) ions. The difference is shown by the two pictures below, the left is copper acetate, the right is hydrated copper sulfate.


In the experiment, described in this web page, it is investigated what happens if the simple acetate ion is replaced by similar ions. The following compounds are made, both in solution and in the solid form:
- copper monochloroacetate
- copper trichloroacetate
- copper propionate
- copper formiate
- copper lactate
It is interesting to see how properties are changing with these anions, which are deviations from acetate.
![]()
![]() Required
chemicals:
Required
chemicals:
-
basic copper carbonate
-
acetic acid
-
monochloro acetic acid
-
trichloro acetic acid
-
propionic acid
-
formic acid
-
lactic acid (either D, L or racemic mix D/L is suitable)
![]() Required
equipment:
Required
equipment:
- test tubes
- means of safely heating a test tube
![]() Safety:
Safety:
- All acids, used in this experiment, are corrosive at high concentrations.
- Monochloro acetic acid and trichloro acetic acid are toxic.
![]() Disposal:
Disposal:
- The waste must be disposed of in a proper waste processing facility. Copper(II) waste in somewhat larger quantities is toxic for aquatic life and so, this waste must not be flushed down the drain.
![]()
Preparation of solutions of the different copper compounds
Here, it is described how each of the compounds can be made from the chemicals, listed above.
Preparation of copper acetate
Making copper acetate can be done by taking 1 ml of glacial acetic acid and mixing this with 3 ml of water, such that appr. 25% of acid is obtained. To this, some basic copper carbonate is added. The liquid will start fizzling and the solid slowly dissolves. Some heating is required and excess acetic acid is required to really dissolve all of the basic copper carbonate. The final result is a dark green/blue solution of copper acetate, in excess dilute acetic acid. It is difficult to get a solution, completely free of solid copper acetate. This can be done, but then the solution must be diluted further to 10 ml.
Preparation of copper monochloroacetate
Making copper monochloroacetate is easier than making copper acetate. Take a spatula full of monochloroacetic acid and dissolve this in 2 ml of water. Add some basic copper carbonate. The basic copper carbonate quickly dissolves with strong fizzling and a clear sky-blue solution is obtained. When fizzling slows down, slightly heat the liquid. Quite a lot of basic copper carbonate can be added and the blue solution can be made rather concentrated. Assure that a slight excess of basic copper carbonate is added. Decant the clear blue solution from the excess of basic copper carbonate, when the fizzling (almost) comes to a halt.
Preparation of copper trichloroacetate
Making this works exactly the same as with the monochloroacetate. Substitute trichloroacetic acid for the monoacetic acid and proceed in the same way. The trichloroacetate also forms a sky-blue solution and this also can be made rather concentrated.
Preparation of copper propionate
Copper propionate is somewhat harder to make. In order to make this, take approximately 1 ml of propionic acid and mix this with 5 ml of water. Add a pinch of basic copper carbonate and gently boil/reflux the liquid for a long time. It is hard to get a clear solution, the solubility of copper propionate is quite low. Even with boiling, there always is a fine crystalline precipitate, which has a bright cyan color. After the boiling, let the liquid stand for a few days in a wide beaker, covered by a piece of glass. During those days, a crystalline crust is formed at the bottom, under a layer of a blue/green solution and any remains of basic copper carbonate, which may be trapped in the fine particles will have reacted after those days.
Preparation of copper formiate
Making copper formiate can easily be done by taking 1 ml of 85% formic acid and mixing this with 3 ml of water, such that appr. 20% of acid is obtained. To this, some basic copper carbonate is added. The liquid will start fizzling and the solid easily dissolves. Some heating speeds up the reaction. One can best keep a slight excess of the acid. The final result is a clear sky-blue solution of copper formiate, in excess dilute formic acid. As with the trichloroacetate and monochloroacetate, rather concentrated solutions can be made without precipitation of solid matter.
Preparation of copper lactate
Preparation of the lactate requires a little more care. It is hard to get all acid converted. One can dissolve some lactic acid (0.5 ml of the 80% acid) in 3 ml of water and add a fairly large excess of basic copper carbonate. At the end of the reaction, the basic copper carbonate only slowly and incompletely dissolves. The solution then is gently boiled for half an hour and every few minutes, a few drops of distilled water are added to compensate for the loss of water. The solution then is allowed to cool down. A sky-blue solution is obtained, with still some solid basic copper carbonate on the glass walls of the test tube. The liquid really needs to be filtered in order to get a clear solution of copper lactate.
![]()
Some results
The picture below shows two test tubes. At the left a solution of copper acetate is shown, which contains a lot of crystalline material. At the right a solution of copper monochloroacetate is shown, with a slight amount of basic copper carbonate at the bottom, and one can see faint bubbling from this basic copper carbonate.

When viewed from below, one can see the dark solid copper(II) acetate in the left test tube and the small amount of excess basic copper carbonate.

The solution of copper propionate looks very similar to the solution of copper acetate. It also is deep blue/green. The picture shows a fairly dilute solution, the rest is at the bottom of the beaker in the form of a fine crystalline precipitate.

No picture was made of the copper trichloroacetate, copper formiate and copper lactate in solution, these look exactly the same as a solution of copper monochloroacetate.
It is interesting to see that the chlorinated acetates, the formiate and the lactate do not form the characteristic dark green/blue solutions, which are formed with plain acetate and with propionate.
![]()
Preparation of the solid compounds by simple evaporation
The liquids, described in the section above, are the following:
- copper acetate, in excess of dilute acetic acid
- copper monochloroacetate, nearly neutral
- copper trichloroacetate, nearly neutral
- copper propionate, in excess of dilute propionic acid
- copper formiate, in excess of dilute formic acid
- copper lactate, hopefully without any excess acid
Preparation of the solid compounds is done by pouring the liquids in a petri dish (assuring that any excess basic copper carbonate is left behind) and allowing the liquid to evaporate in one day. The petri dishes were placed in a warm place (appr. 50 şC) for one day. After this day, a solid crust is formed, and this crust is scraped from the glass of the petri dish and then the solids are allowed to dry for another 6 hours in a warm place. After this treatment, completely dry solids are obtained.
As expected, the copper acetate and copper propionate form dark green/blue solids, with colors very much like the color of the solutions. The copper trichloroacetate forms a sky-blue solid, like copper sulfate. Given the color of its concentrated solution, this could be expected. The most remarkable result is obtained for the monochloroacetate. Although the solution is blue like a copper sulfate solution, the solid is dark green/blue, like plain copper acetate and copper propionate.
Copper formiate forms a nice crystalline solid of a remarkably pale blue color. It is much lighter than the solid copper trichloroacetate. Copper lactate does not form a crystalline solid, but an amorphous mass, which has colors, ranging from sky-blue to almost white, depending on the roughness of the surface of the solid.
Some pictures
The solids were collected and put in small vials. These vials are shown in the picture below, which shows from left to rigt copper acetate, copper monochloroacetate and copper trichloroacetate.

Another comparative picture is made, in which the three blue compounds can be compared. The following picture shows from left to right copper lactate, copper formiate and copper trichloroacetate.

There is a slight color difference between the copper acetate and the copper monochloroacetate. This difference could be due to a slight color difference of the compound, but it could also be due to the finer division of part of the monochloroacetate. The acetates can be made into a very fine powder, and this fine powder has a much lighter color, it is bright cyan.
All solids are grouped in the following picture, at the left the formiate, in the middle, the acetate, monochloroacetate and trichloroacetate, and at the right the propionate and lactate.

Copper formate forms interesting crystals, even when evaporated from a petri dish in just a few hours. Click here for the pictures. They are not part of this web page, because that distracts from the purpose of this webpage, in which the coordination properties of the different anions are compared.
![]()
Discussion of results
This experiment shows that copper acetate and copper propionate form a deep green/blue complex in solution, and in the solid form. According to an internet search, the net formula for copper propionate is (CH3CH2COO)2Cu·H2O and this is very similar to the formula for copper acetate. Combining this data with the observations from the experiment, there is strong evidence for a structure of copper propionate, which is similar to the structure of copper acetate, with the two oxygen atoms connected to the two central copper atoms.
Copper monochloroacetate only forms the deep green/blue complex in the solid form, its solution in water is blue, like a solution of copper sulfate. Even very concentrated solutions of the monochloroacetate are sky-blue and not dark green/blue. Copper trichloroacetate also in the solid form does not form the deep green/blue complex anymore and the solid form most likely is a plain salt with hydrated copper(II) ions and free trichloroacetate ions in the crystal lattice.
The chlorine atoms make the complex with copper(II) atoms more labile. Without chlorine atoms, the complex is sufficiently stable, that it persists, even in dilute aqueous solution. With one chlorine atom, the complex only is stable when there is no free water (apparently the water ligands replace the monochloroacetate ligands, but they are not attached so firmly that they cannot escape when the solid is dried). With three chlorine atoms, the complex cannot be formed at all anymore.
All observations can nicely be explained by the so-called inductive effect of attached groups on a carbon atom.
If taking acetate as reference, then we can regard propionic acid, monochloroacetic acid, trichloroacetic acid and lactic acid as substitution products with H atoms on the CH3-group replaced by something else:
- propionic acid, one H-atom replaced by CH3-group. Relative to the replaced H-atom, the CH3 group is slightly electron releasing.
- monochloroacetic acid, one H-atom replaced by a Cl-atom. A Cl-atom is rather electron withdrawing.
- trichloroacetic acid, all three H-atoms replaced by a Cl-atom. These three Cl-atoms together are strongly electron withdrawing.
- lactic acid, one H-atom replaced by a CH3-group, another H-atom replaced by a OH-group. The electron withdrawing effect of the OH group is the dominant effect.
The structure of the acetate ion is as follows:
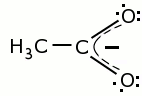
What is important is the two lone pairs on the oxygen atoms. These are used in coordinate bonds with the two copper atoms, as shown at the start of this webpage. The acetate ion easily shares one lone pair per oxygen atom with a central metal ion, allowing easy formation of complexes. If, however, one or more of the H-atoms on the CH3-group are replaced by strongly electron-withdrawing groups (such as Cl, OH), then the electron pairs on the oxygen atoms cannot easily be shared anymore, and complex formation becomes more difficult. This is exactly what is observed, and one even can see that replacement of all H-atoms by Cl-atoms makes complex formation more difficult than replacement of a single atom. The acetate and propionate ions have similar coordinating strength, the other ions have less coordinating strength.
The fact that formiate ion does not form a deep green/blue complex can also be explained. Now the complete CH3-group is replaced by a single H-atom. A CH3-group is more electron releasing than a H-atom. The effect now is much stronger than when going from acetate to propionate, and this is because the replacement now is directly on the C-atom, which has the two O-atoms attached.