


Burning gases -- different flame colors
This is a simple series of experiments, which demonstrates that different flammable gases show different flame colors when they are burnt.
The gases are either used directly or made by means of a simple chemical reaction. The gases are sucked in a syringe of 20 ml, to which a glass tube is attached with a sharp tip. By simply pressing the gas out of the syringe and keeping it near a flame of a small alcohol burner, the gas from the syringe is ignited. The flame is maintained by pressing the gas slowly out of the syringe. At the end of each experiment the gas is pressed out of the syringe somewhat faster.
This webpage contains several experiments, which all can be performed independently. If one does not have the chemicals for one particular experiment, then the others still can be done if the materials for that are available. The chemicals in small font are needed for making the gas, under which they are mentioned.
![]()
![]() Required
chemicals:
Required
chemicals:
-
butane
-
methane (natural gas)
-
hydrogen
-
magnesium
-
dilute hydrochloric acid
-
-
carbon monoxide
-
formic acid, 80% or better
-
concentrated sulphuric acid
-
-
cyanogen
-
sodium cyanide
-
copper sulfate
-
-
methyl nitrite
-
potassium nitrite
-
methanol
-
dilute hydrochloric acid
-
![]() Required
equipment:
Required
equipment:
-
test tube
-
syringe
-
glass tube with sharp tip
-
little flexible plastic tube
![]() Safety:
Safety:
Safety is discussed in the individual experiments for the different gases.
![]() Disposal:
Disposal:
- The production of cyanogen uses copper sulfate and sodium cyanide. Besides the gas, impure copper(I) cyanide is produced. This must be neutralized by adding excess bleach to it and then it can be acidified. The resulting solution can be flushed down the drain. Only a very small amount of copper sulfate is used, not worth the effort of bringing it to a waste processing facility.
![]()
Butane gas
Butane gas simply is taken from a cigarette lighter refill canister. Just press a small amount of gas into a clean test tube and then suck the gas in a syringe. Use a little flexible tube, attached to the tip of the syringe, such that the gas is sucked in from the bottom of the test tube.
 The
gas burns with a bright flame, which at the bottom is faint blue. The bright
flame is due to solid particles of carbon, which are formed in the
oxygen-deficient central part of the flame due to pyrolysis and which start glowing (emitting
black body
radiation). At a greater height in the flame, the solid particles are burnt
again and no soot is produced. A
video is present of the burning of the gas, it
has a download size of 897 kByte. When the gas is pressed out of the syringe at
a higher speed, then the flame becomes much larger, and some soot is observed.
Compared to many other gases, the flame of butane is much brighter. Black body
radiation is much more intense than the glow of excited molecules and ions
falling back to a lower energy state, which is responsible for
the weaker flame colors, like the blue at the bottom part of the butane-flame.
The
gas burns with a bright flame, which at the bottom is faint blue. The bright
flame is due to solid particles of carbon, which are formed in the
oxygen-deficient central part of the flame due to pyrolysis and which start glowing (emitting
black body
radiation). At a greater height in the flame, the solid particles are burnt
again and no soot is produced. A
video is present of the burning of the gas, it
has a download size of 897 kByte. When the gas is pressed out of the syringe at
a higher speed, then the flame becomes much larger, and some soot is observed.
Compared to many other gases, the flame of butane is much brighter. Black body
radiation is much more intense than the glow of excited molecules and ions
falling back to a lower energy state, which is responsible for
the weaker flame colors, like the blue at the bottom part of the butane-flame.
![]()
Methane gas
The methane gas is taken from the kitchen gas stove. The natural gas, used for cooking, mostly is methane gas in most countries, with some nitrogen mixed in. The mixed in nitrogen is not a real issue, it hardly affects flame color.
Take a test tube, and keep it inverted over one of the gas burners on the stove, at the place, where no air is mixed in. It might be necessary to take off part of the gas burner in order to do so. Keep the test tube over the burner for a few seconds while the gas is flowing. Turn off the gas flow and quickly suck in the gas from the bottom of the test tube, while keeping the test tube upside down (so the bottom is the highest point of the test tube). Methane gas has a lower density than air, hence the need to keep the test tube upside down.
 The
flame of the burning methane resembles that of the burning butane, but it is
smaller (for the same rate of flow of gas). The different flame sizes can easily
be explained. Per volume of gas, butane has 4 times as much carbon and 2� times
as much hydrogen. So, there is much more fuel in the same volume of butane gas.
The
flame of the burning methane resembles that of the burning butane, but it is
smaller (for the same rate of flow of gas). The different flame sizes can easily
be explained. Per volume of gas, butane has 4 times as much carbon and 2� times
as much hydrogen. So, there is much more fuel in the same volume of butane gas.
The video of burning methane gas has a download size of 625 kByte. Again, as with butane gas, when the gas is pressed out of the syringe at a higher speed, the flame becomes much larger.
![]()
Carbon monoxide
Carbon monoxide is easily made by putting 0.5 ml of formic acid (80% or better) and 2 ml of concentrated sulphuric acid in a test tube and gently heating it, while keeping it loosely stoppered. When production of gas becomes much slower, then quickly remove the stopper and immediately suck the gas from the test tube into a syringe. Carbon monoxide is slightly less dense than air, but if the action of sucking the gas in the syringe is done sufficiently fast, then this perfectly works.
![]() Beware, carbon monoxide is very toxic, odorless and colorless without any
warning. Make the carbon monoxide in a well-ventilated room, under an open
window, or a suitable fume exhaust. In a large well-ventilated room the risk of
making carbon monoxide from only 0.5 ml of formic acid is very limited. Do not scale up, if you are
not confident that your ventilation is good enough.
Beware, carbon monoxide is very toxic, odorless and colorless without any
warning. Make the carbon monoxide in a well-ventilated room, under an open
window, or a suitable fume exhaust. In a large well-ventilated room the risk of
making carbon monoxide from only 0.5 ml of formic acid is very limited. Do not scale up, if you are
not confident that your ventilation is good enough.
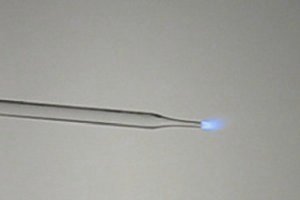 Carbon
monoxide burns with a small beautiful blue flame. There is no formation of solid
particles like in methane and butane, hence the flame has much lower light
output. Its color is due to photon emission of excited molecules and ions in the
very hot gas mix, which fall back to a lower energy state.
Carbon
monoxide burns with a small beautiful blue flame. There is no formation of solid
particles like in methane and butane, hence the flame has much lower light
output. Its color is due to photon emission of excited molecules and ions in the
very hot gas mix, which fall back to a lower energy state.
The video of burning carbon monoxide gas has a download size of 681 kByte. When the gas is pressed out of the syringe at a high speed, then a very nice effect can be observed. The flame is moving away from the tip of the glass tube and can easily reach a distance of several cm. At a certain point, the flame disappears.
![]()
Hydrogen
Hydrogen easily is made by taking two test tubes. Put 5 ml of dilute hydrochloric acid (10%) in a test tube, and throw in a few cm of magnesium ribbon. Immediately stopper the test tube with a stopper, which has one hole in it and a tube going through it. Collect the gas in another test tube, which is held upside down according to the following drawing:
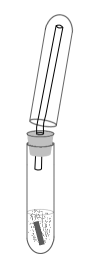
When the piece of magnesium is dissolved almost completely and production of gas becomes slower, then quickly suck the gas from the upper test tube in a syringe, while keeping it upside down and keeping the other tube in it as well, from which still some gas is flowing. In this way, fairly pure hydrogen gas is collected without much air mixed in.
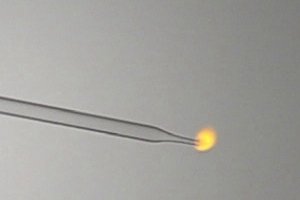 Hydrogen
gas burns with a small orange flame, which goes around the tip of the glass
tube. This little flame is not like the orange flame of butane or methane, but
it really is due to photon emission of excited hydrogen atoms, which produces orange light. No solid
particles are formed in the hydrogen flame. The
video
of burning hydrogen gas has a download size of 553 kByte. When the gas is
pressed out of the syringe at a high speed, then the flame becomes larger, and
at sufficiently high speed a popping noise is produced. The gas mixes with air
and then this mix at once ignites. This popping noise, however, is not created
in all cases. In many other cases, there simply is a larger orange 'cloudy'
flame around the tip of the glass tube.
Hydrogen
gas burns with a small orange flame, which goes around the tip of the glass
tube. This little flame is not like the orange flame of butane or methane, but
it really is due to photon emission of excited hydrogen atoms, which produces orange light. No solid
particles are formed in the hydrogen flame. The
video
of burning hydrogen gas has a download size of 553 kByte. When the gas is
pressed out of the syringe at a high speed, then the flame becomes larger, and
at sufficiently high speed a popping noise is produced. The gas mixes with air
and then this mix at once ignites. This popping noise, however, is not created
in all cases. In many other cases, there simply is a larger orange 'cloudy'
flame around the tip of the glass tube.
![]()
Methyl nitrite
Mix 1 ml of methanol with 3 ml of dilute hydrochloric acid (10%). In this mix, throw a spatula full of solid potassium nitrite. Gas production starts at once and is fairly vigorous, but not exothermic. Keep the test tube loosely stoppered while gas is produced. When production of gas becomes much slower, then quickly remove the stopper and quickly suck the gas from the test tube into a syringe. Methyl nitrite has a much higher density than air and it easily can be sucked into a test tube when the gas is sucked from just above the surface of the liquid.
Do not use sodium nitrite for this experiment. Tiny droplets of the solution may go into the gas above the liquid and these tiny droplets contain sodium ions. They spoil the flame color and most likely an orange/yellow flame is produced when sodium nitrite is used for making the methyl nitrite.
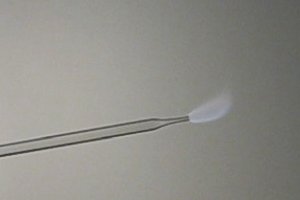 Methyl
nitrite burns with a grey flame. This is a rather unusual flame color.
The intensity of the light from the flame is fairly low.
Methyl
nitrite burns with a grey flame. This is a rather unusual flame color.
The intensity of the light from the flame is fairly low.
The video of burning methyl nitrite gas has a download size of 708 kByte. When the gas is pressed out of the syringe at a high speed, then the flame becomes much larger. The effect is different from the effect with butane gas or methane gas. For the latter two gases, the flame grows both in width and in length, with methyl nitrite, the flame mostly grows in length, its becomes a narrow long flame.
![]()
Cyanogen
Cyanogen can be made by taking a spatula full of copper sulfate 5-hydrate and dissolving this in 2 ml of water. Also take a spatula full of sodium cyanide or potassium cyanide and dissolve this in a ml of water. Heat both solutions. They need not boil, but they should be fairly hot, such as hot tap water. When the two solutions are mixed, then a dirty brown precipitate is formed and a colorless gas. Immediately after mixing the two liquids, loosely stopper the test tube and allow the gas to be produced. Slight heating helps formation of the gas. When production of gas becomes slow, then suck out the gas from the test tube. Some shaking may be necessary before gas is taken out, in order to break the brown foam, which is produced in the reaction.
![]() Cyanogen is
very toxic, its toxicity is comparable to that of hydrogen cyanide.
Make the gas in a fume hood, or do this experiment outside. Also limit
the amount of sodium cyanide to a single spatula of just 200 mg or so.
Do not scale up, if you are not confident that your ventilation is
good enough.
Cyanogen is
very toxic, its toxicity is comparable to that of hydrogen cyanide.
Make the gas in a fume hood, or do this experiment outside. Also limit
the amount of sodium cyanide to a single spatula of just 200 mg or so.
Do not scale up, if you are not confident that your ventilation is
good enough.
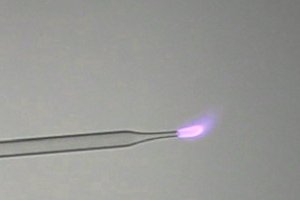 The
flame color of burning cyanogen is the most remarkable of all experiments on
this page. The flame has a beautiful deep saturated rose/pink color with a blue
'halo' around it.
The
flame color of burning cyanogen is the most remarkable of all experiments on
this page. The flame has a beautiful deep saturated rose/pink color with a blue
'halo' around it.
The video of burning cyanogen gas has a download size of 601 kByte. When the gas is pressed out of the syringe at a high speed, then the flame leaves the tip of the glass tube. The effect is even more pronounced than with carbon monoxide. At a certain point, the flame disappears, just as in the case with carbon monoxide.