Carbon - C

Two rods of graphite, each with a diameter of 4 mm. These rods are used as electrodes in batteries and consist of almost pure graphite, being one of the allotropes of carbon. Graphite is a good conductor of electricity, but only in two directions. The conductivity in the direction, perpendicular to these two directions is much less. This is due to the fact that graphite forms large planes of carbon atoms, arranged hexagonally, with one electron in a conductivity band. The rods, shown above, consist of many very small particles of graphite, and as such they are black and not shiny. Their conductivity is the same in all directions. This is due to the fact that all particles of graphite have their own random orientation.
Below, a picture is shown of a piece of almost 100 grams of pure graphite, which is a large "crystal" and not made of many small particles of graphite. It looks shiny and dark grey. It is so soft, that by touching it, one even obtains black fingers. The powder, scraped from this piece of graphite is black. One can draw black lines with this, by moving it along a piece of paper. The conductivity of this piece depends on the direction in which the current flows.

![]()
Another allotrope of carbon is amorphous carbon, which does not conduct electricity at all. Below, a picture is given of a sample of very pure amorphous carbon, in the form of small granules. This allotrope of carbon also is black.

![]()
Yet another allotrope of carbon is diamond. Pure diamond is completely transparent like glass. It is a very hard compound, with all carbon atoms arranged in a 3D lattice, where each carbon atom has neighbor atoms at the vertices of a tetrahedron. The picture below shows many minute artificial diamonds, each diamond having a size of approximately 0.5 mm. These little diamonds are not 100% pure, hence their yellow color.

A close-up of these diamonds is shown below. This close-up nicely shows the faceted sides of the diamonds, and it also shows their transparency.

![]()
A completely new and different allotrope of
carbon was discovered in the 1980s. In 1996 the Nobel prize was awarded
to R.F. Curl, H.W. Kroto and R.E. Smalley for their discovery of the
so-called fullerenes. This is
a whole class of compounds, in which carbon atoms form large but finite
structures. The most common structures look like footballs or like
tubes.
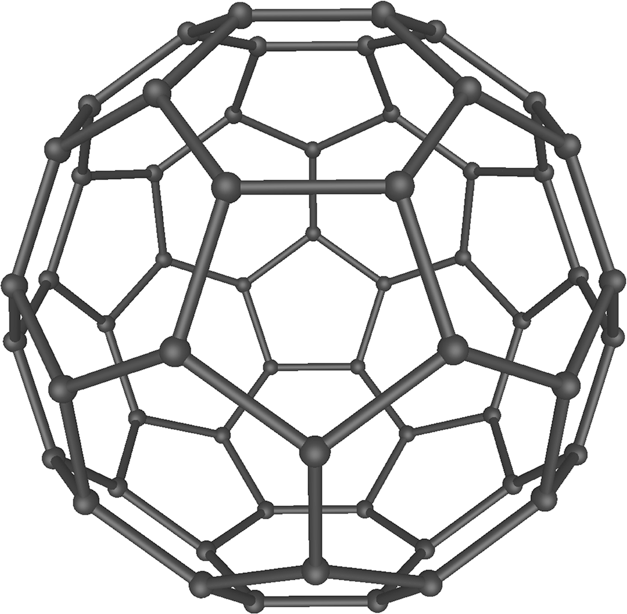
The best-known representative of this class of compounds is C60,
which is depicted in the picture at the left. It contains 60 carbon
atoms, connected to each other in the same way as the vertices of the
leather parts of a football (soccerball). Soon, it became clear that C60
is just one representative of a whole class of compounds. Nowadays many
similar compounds are known, some of them having spherical shapes like C60,
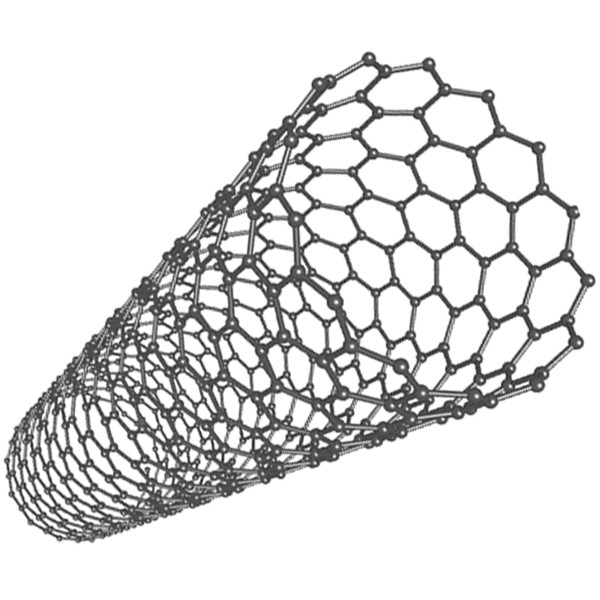
but also egg-shaped compounds are known and tube-like compounds are
known, both with open ends and closed ends. The next best-known
compound is C70, which has an egg-like shape.
These compounds form discrete molecules and in this they differ from the 'classic' allotropes of carbon. As a consequence, many of the fullerenes are soluble in non-polar solvents, albeit only very sparingly so, in the order of a mg or tens of mg per 100 ml. Despite the low concentration, such solutions have intense colors.
The fullerenes are dark crystalline compounds, black, or dark brown. The picture below shows 200 mg of C60.

This dark brown compound dissolves in naphtalenes, tetralin, carbon disulfide, xylene, toluene, trichloroethene and many other non-polar solvents. Such solutions are purple. Best results are obtained with naphtalenes and tetralin, next best results can be obtained with carbon disulfide. Carbon disulfide allows dissolving tens of mg in 100 ml of liquid. Below, a picture is shown of an ampoule, which contains a few ml of a saturated solution of C60 in carbon disulfide.

The picture below shows the saturated solution in more detail. The dark specks are small particles of undissolved C60. In making this ampoule, a small excess amount of C60
was used so that a saturated solution would be obtained and a small
quantity of solid material remains. The color of such solutions is
remarkably intense, taking into account the low concentration of these
solutions.

Other fullerenes also are soluble in non-polar solvents, e.g. C70 gives wine-red solutions and there are also fullerenes, which give brown or yellow solutions.
At the moment, a lot of research is done on fullerenes. The closed spherical or egg-shaped compounds can hold small molecules or atoms inside them, and special molecules are made already, like C60·K and C60·Xe, with the K or Xe atom trapped inside the sphere of carbon atoms. The tube-shaped compounds (a.k.a. nanotubes) may have applications in future electronics. The tubes can be made so narrow that trapped electrons obtain very special and interesting properties, which only can be understood by using quantum mechanical descriptions.
Fullerenes are available to the general
public, but using them for chemical experiments unfortunately is beyond
reach of most people, due to their cost. The cheapest fullerene (C60)
has a price tag of appr. $0.10 per mg, other fullerenes easily go
beyond $1 per mg. The last few years, the price of some tube-shaped
fullerenes tends to go down somewhat.